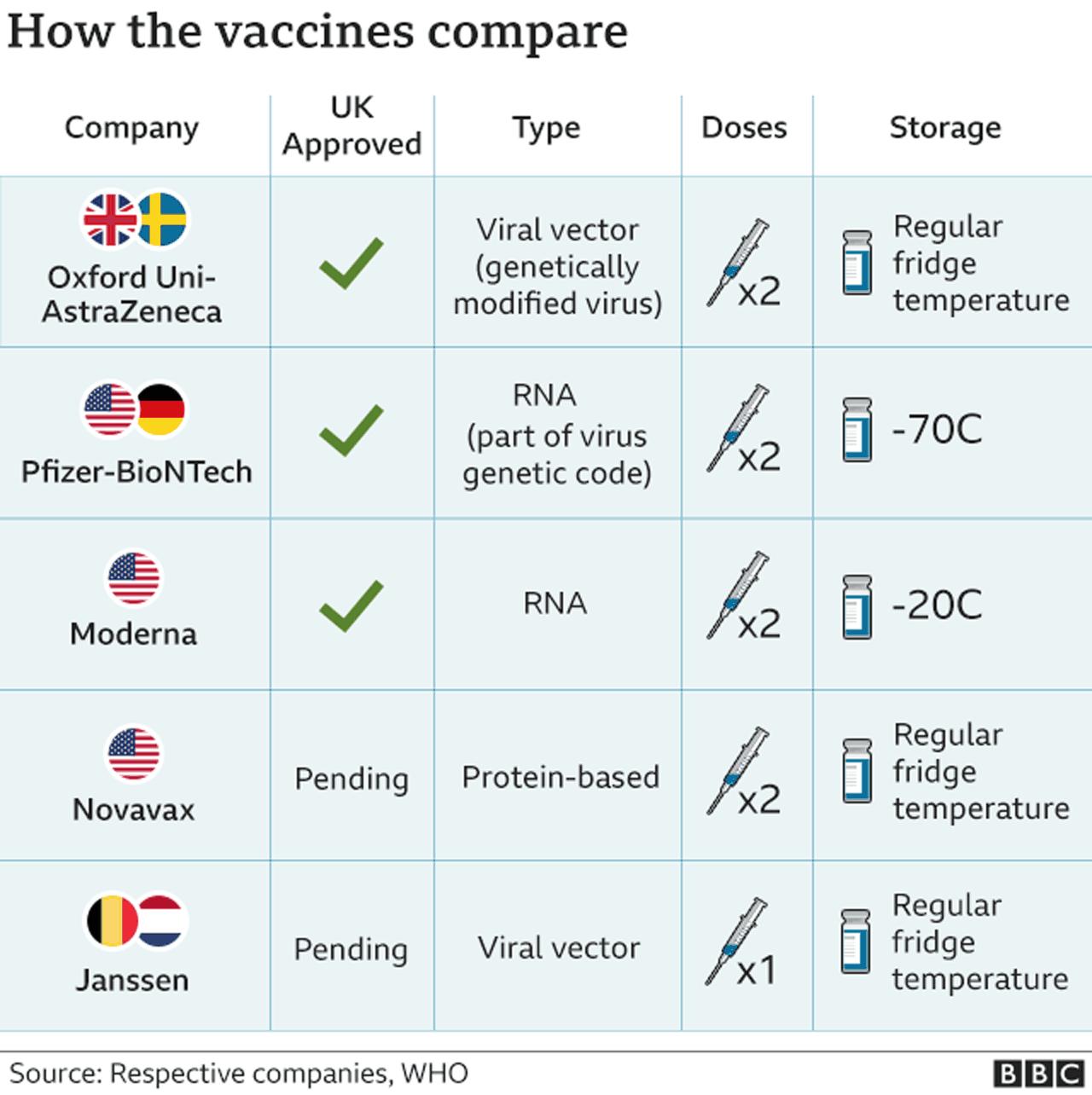 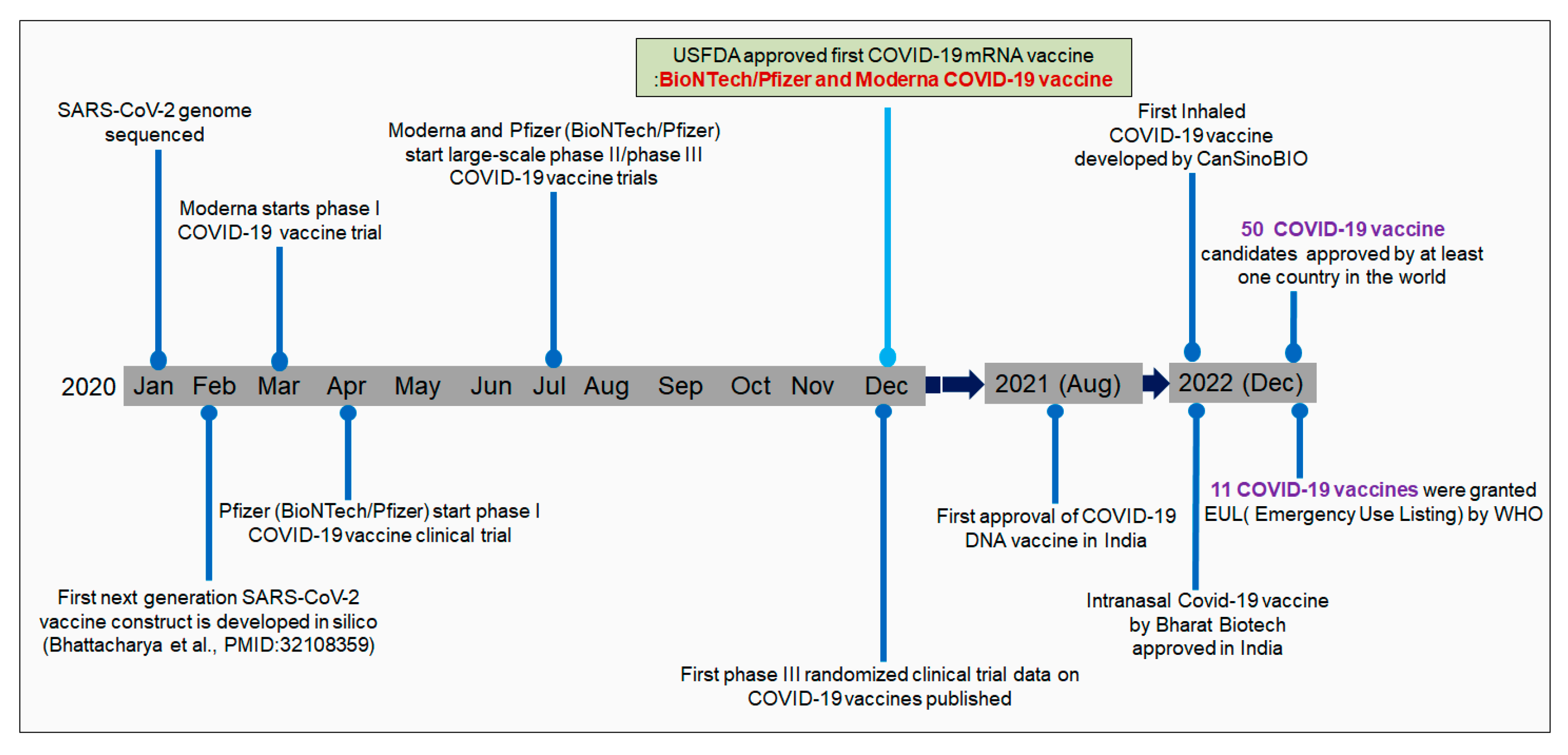 
Nevertheless, the announcement raised concerns about undue political influence over the release of a COVID-19 vaccine, leading the chief executives of nine pharmaceutical companies to publicly pledge to “make the safety and well-being of vaccinated individuals” their “top priority,” while stating that they would only seek approval of a vaccine if a Phase 3 trial establishes that it is both safe and effective. Whether this vaccine would be granted official licensure from the Food and Drug Administration (FDA) or be released by an emergency use authorization (EUA)-a tool by which the FDA can authorize use of medical products, including vaccines, that have not completed clinical trials-is unclear. This fear was stoked in early September, when the Centers for Disease Control and Prevention (CDC) told state officials across the country to prepare for the distribution of a COVID-19 vaccine by November 1, just days before the presidential election. What’s more, according to the report, when asked about the pace of the vaccine approval process, 78% say their primary concern "is that it will move too fast, without fully establishing safety and effectiveness.” According to a survey conducted by the Pew Research Center, the number of Americans who planned to get a COVID-19 vaccine has fallen from 72% in May to just 51% in September. That leaves many Americans concerned about how safe a COVID-19 vaccine will be. Yet, because of the urgency created by this global pandemic-and the serious illness and death rates associated with COVID-19-preclinical and clinical trials to test the efficacy and safety of COVID-19 vaccine candidates are happening at a rapid, almost frenetic, pace. (In fact, the fastest vaccine ever developed-a mumps vaccine-still took four years.) In March, at the start of the COVID-19 pandemic, there was a consensus among health care providers and public health officials that a vaccine that provided complete immunity to SARS-CoV-2, the virus that causes COVID-19, would effectively end the pandemic.Īt the time, experts suggested that the development of a safe and effective COVID-19 vaccine could be accomplished in 12 to 18 months, even though vaccine development typically takes about 10 years. Because information about COVID-19 changes rapidly, we encourage you to visit the websites of the Centers for Disease Control & Prevention (CDC), World Health Organization (WHO), and your state and local government for the latest information. Halsey has authored more than 250 peer reviewed publications, 6 books, and 43 book chapters on the prevention and treatment of infectious diseases through vaccination, and he has participated in the development of more than 100 guidelines for the use of vaccines while serving on advisory groups for the WHO Expanded Programme on Immunization, the Advisory Committee for Immunization Practices (ACIP) for CDC, and the American Academy of Pediatrics Committee on Infectious Diseases.ĭr Halsey has focused his research and teaching in recent years on vaccine safety.Note: Information in this article was accurate at the time of original publication. After 5 years as a faculty member at Tulane University he has been at Johns Hopkins University since 1985.ĭr. Halsey served in the Epidemic Intelligence Service at the Centers for Disease Control and Prevention(CDC) in Atlanta. 
Halsey, MD, Director Emeritus of IVS, is an Emeritus Professor in the Department of International Health at the Johns Hopkins Bloomberg School of Public Health and in the Department of Pediatrics in the School of Medicine.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
